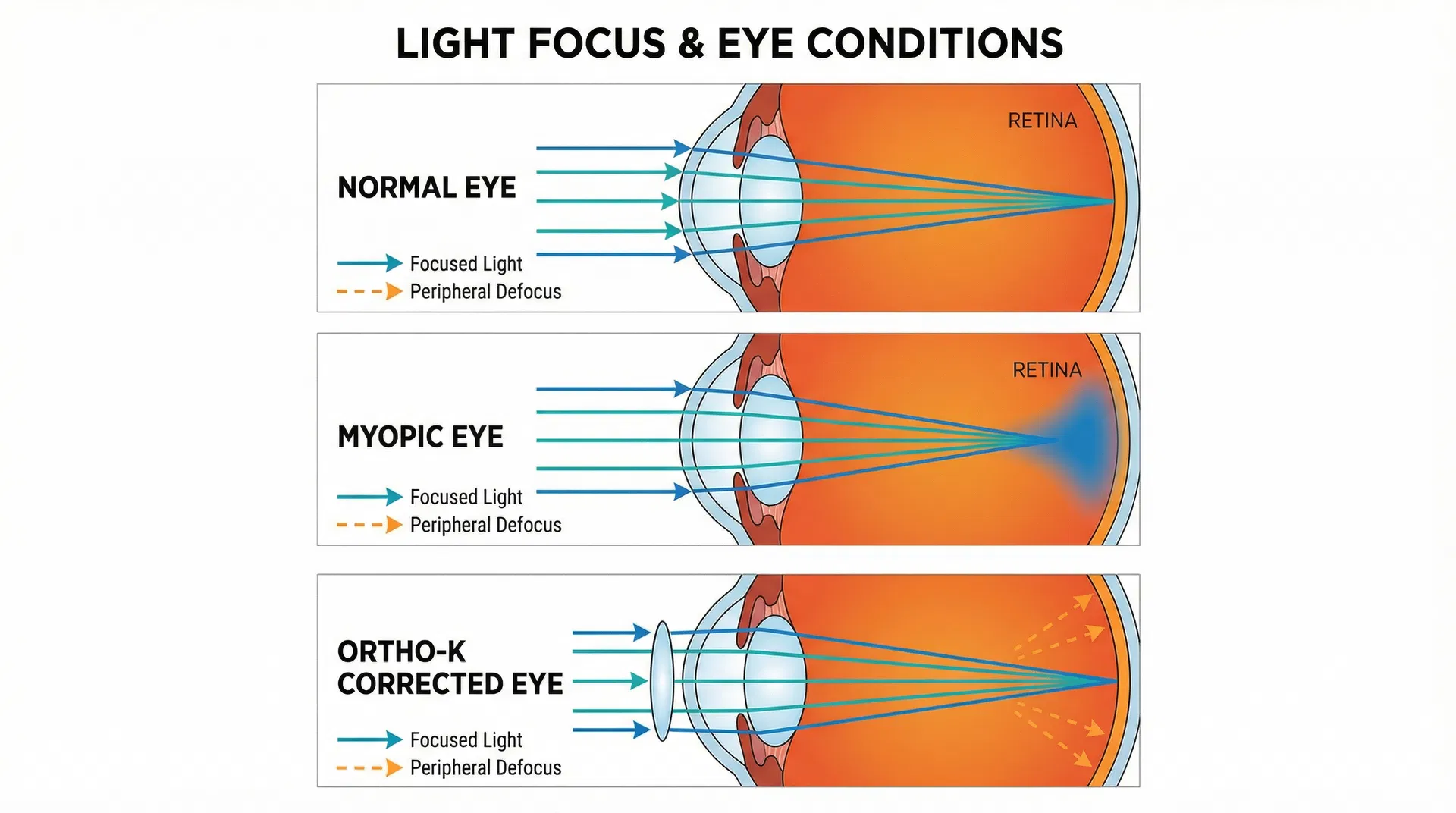
As an optometrist who has dedicated my career to fighting childhood myopia, I’ve had the privilege of helping countless families in Fayetteville and across Cumberland County discover the life-changing benefits of orthokeratology. It’s a technology I’m deeply passionate about, not just for its ability to provide clear vision without daytime glasses or contacts, but for its proven effectiveness in myopia control. But with any medical treatment, especially one for our children, the first question is always—and rightly so—"Is it safe?"
I hear it from parents every day. They’ve heard about ortho-k, maybe from a friend or through their own research, and they’re intrigued. The idea of their child being free from glasses, especially for sports and swimming, is incredibly appealing. But the thought of a contact lens worn overnight can be intimidating. That’s why I want to pull back the curtain and talk about the single most important factor in ensuring the safety and efficacy of ortho-k: FDA approval.
In This Article
- What FDA Approval Means for Your Family’s Safety
- A Landmark Achievement: The History of FDA Approval for Myopia Control
- Behind the Approval: The Rigorous Testing Process
- Post-Market Surveillance: The Safety Watch Doesn’t End
- How to Verify Your Lenses are FDA-Approved
- Your Partner in Safe Myopia Control
- Frequently Asked Questions (FAQs)
What FDA Approval Means for Your Family’s Safety
So, what does it really mean when we say an ortho-k lens is "FDA-approved"? The U.S. Food and Drug Administration (FDA) is the federal agency responsible for protecting public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological products, and medical devices. And yes, contact lenses, including ortho-k lenses, are classified as medical devices.
For a device to earn FDA approval, it must go through a rigorous, multi-year process of scientific and clinical review. The manufacturer has to provide extensive data from laboratory studies and human clinical trials to prove two fundamental things:
- It is safe: The device does not pose an unreasonable risk of illness or injury.
- It is effective: The device performs as promised and delivers a real clinical benefit.
This isn’t just about paperwork. It’s about a deep, scientific evaluation of the device’s design, materials, manufacturing processes, and performance in real patients. The FDA’s review is exhaustive, and their standards are among the highest in the world. When you choose an FDA-approved ortho-k lens, you’re not just getting a piece of plastic; you’re getting a medical device that has been scrutinized, tested, and validated to meet stringent safety and performance benchmarks. It’s a seal of approval that provides peace of mind, and frankly, it’s a non-negotiable for me as a practitioner.
Think of it like this: you wouldn’t give your child a medication that wasn’t approved by the FDA. The same standard should apply to anything you put in their eyes. This approval is the bedrock of patient safety in the world of orthokeratology.
A Landmark Achievement: The History of FDA Approval for Myopia Control
Ortho-k's journey to becoming a trusted option for myopia control is marked by key FDA milestones. Previously, it was an "off-label" treatment, lacking the safety assurances of formal FDA approval.
In 2002, the FDA approved Paragon CRT®, the first lens specifically for overnight orthokeratology. This validated the treatment as a safe and effective method for vision correction.
Euclid's Emerald™ lens followed in 2004, and other designs have since been approved, further establishing the safety of overnight orthokeratology.
Here is a look at some of the leading FDA-approved ortho-k lenses available in the United States today:
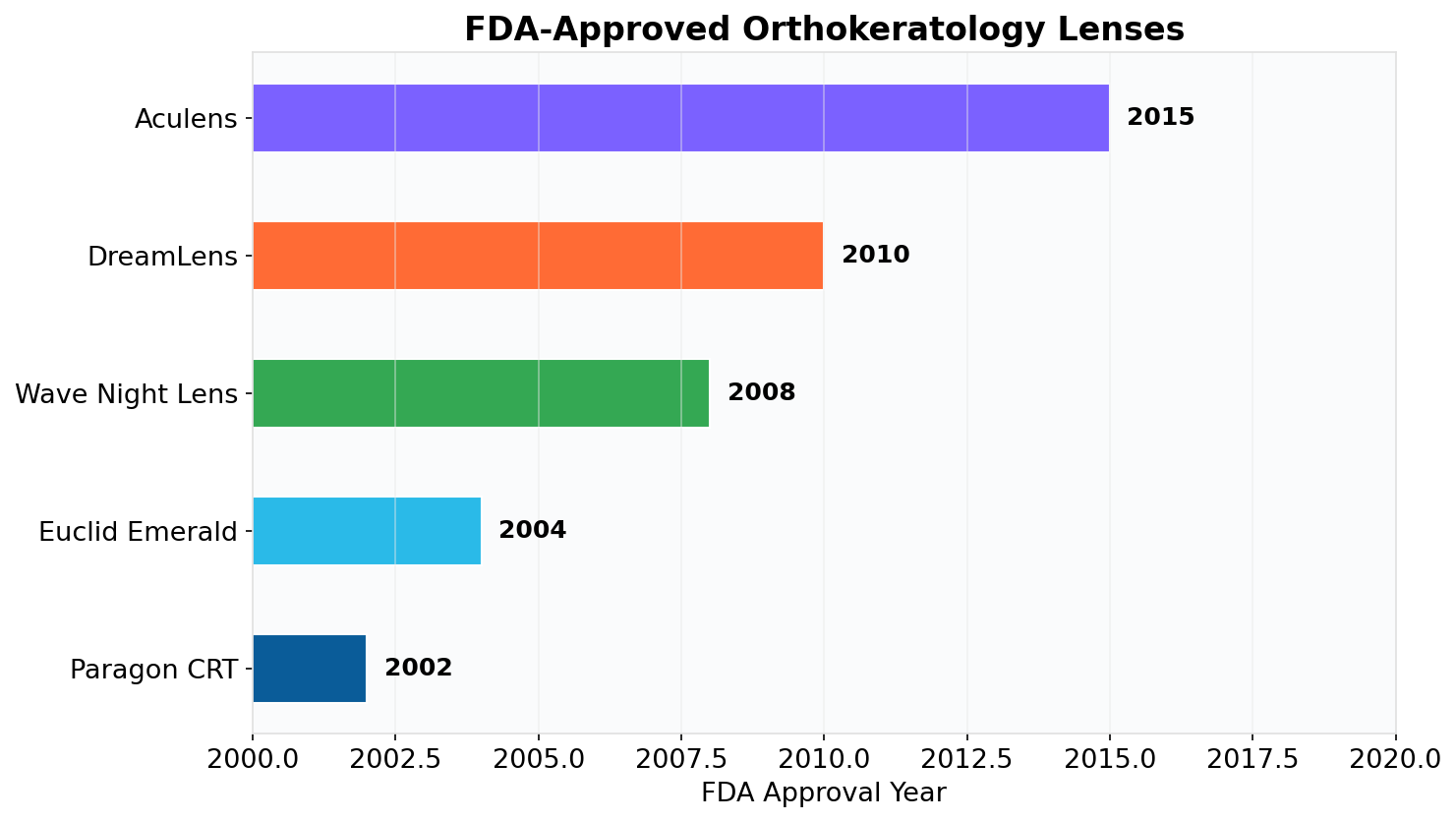
This chart showcases some of the most trusted and widely used FDA-approved orthokeratology lens designs in the U.S. market.
This history demonstrates a commitment to safety and innovation, transforming ortho-k into a powerful tool for myopia control.
Behind the Approval: The Rigorous Testing Process
The FDA approval process is a marathon, involving several stages of scrutiny.
Pre-clinical studies test the lens material's oxygen permeability, durability, and resistance to deposits. Computer simulations model the lens's interaction with the cornea.

In state-of-the-art labs like this one, every aspect of an ortho-k lens is meticulously tested to ensure it meets the highest standards for safety and quality before it ever reaches a patient’s eye.
Successful pre-clinical data leads to human clinical trials involving hundreds of patients. Investigators monitor patients closely over an extended period.
During these trials, we collect a mountain of data on:
- Efficacy: Does the lens successfully reduce myopia and provide clear vision throughout the day?
- Safety: Are there any adverse events, such as inflammation, infection, or corneal staining? How does the eye health of the trial participants compare to a control group?
- Fit and Comfort: How well does the lens fit on a variety of corneal shapes? Is it comfortable for overnight wear?
The comprehensive data is submitted to the FDA for review by a team of experts. Approval is granted only when the benefits are proven to outweigh the risks.
Post-Market Surveillance: The Safety Watch Doesn’t End
FDA approval isn't the end of the safety story. Post-market surveillance is a critical phase where the FDA continues to monitor the performance of lenses and manufacturers must report any serious adverse events.
This feedback loop allows the FDA to take action if issues arise, from updating labels to recalling products, forming a crucial safety net for patients.
Parents often ask about infection risks. While all contact lenses carry some risk, studies show that with proper hygiene, the risk of microbial keratitis with FDA-approved ortho-k lenses is very low and comparable to other overnight lenses.
Let’s look at the data:
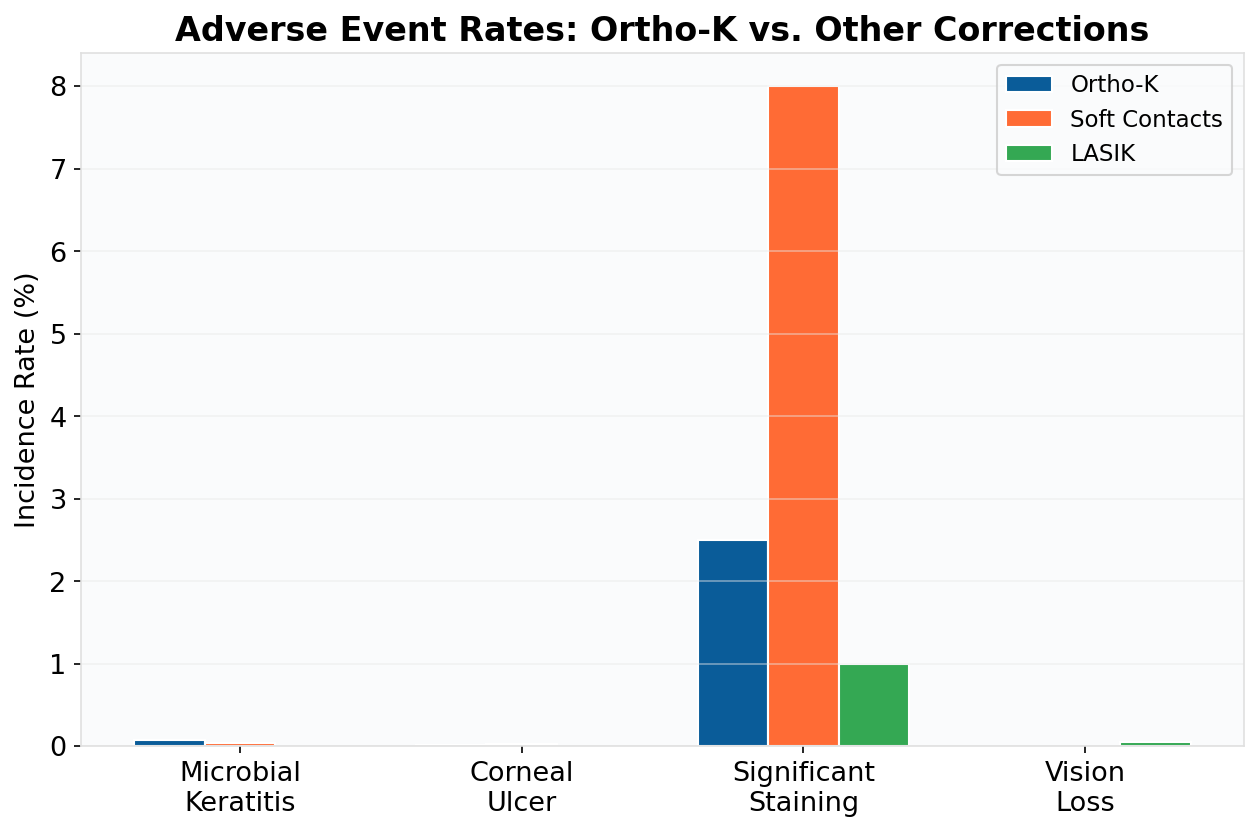
This chart compares the estimated incidence of microbial keratitis, a rare but serious complication, across different types of contact lenses. As you can see, the rate for ortho-k is similar to other approved overnight modalities and remains very low overall. [1]
For parents, this means that FDA-approved ortho-k has a strong safety profile. Minimizing risk comes down to choosing an experienced provider and following care instructions diligently. At our Fort Liberty area clinic, we prioritize patient education to ensure every family is confident in their lens care routine.
How to Verify Your Lenses are FDA-Approved
You have the right to know what device is being used. To ensure you're getting an FDA-approved lens, simply ask your doctor.
A reputable practitioner will readily provide the brand and design of the lens and confirm its FDA approval. You can also check the packaging for the FDA label.
Here’s a quick comparison of some of the leading FDA-approved ortho-k lens brands available today:
| Lens Brand | Manufacturer | Year of FDA Approval | Key Features |
|---|---|---|---|
| Paragon CRT® | CooperVision Specialty EyeCare | 2002 | The first FDA-approved overnight ortho-k lens; widely used with a long track record of success. |
| Euclid Emerald™ | Euclid Systems Corporation | 2004 | Known for its advanced lens design software and customization options. |
| BE-Retainer | Bausch + Lomb | 2021 | A more recent addition, offering advanced material technology for high oxygen permeability. |
| GOV® (Global Orthokeratology Vision) | Global-OK Vision | 2015 | Offers a wide range of parameters to fit a variety of patients. |
For further verification, you can search the FDA's public database of approved medical devices. At sleepSEE®, we exclusively use FDA-approved lenses from leading manufacturers as part of our commitment to the highest standard of care for our Fayetteville patients.
Your Partner in Safe Myopia Control
Choosing to start your child in a myopia control program is a big decision. It’s an investment in their future, helping to protect their vision and reduce their risk of serious eye diseases later in life. The safety of that treatment is, and always should be, the number one priority.
The FDA approval process provides a powerful, evidence-based framework for ensuring that safety. It’s a rigorous, scientific, and ongoing process that ensures the ortho-k lenses we use are both safe and effective. It’s the reason I can confidently recommend this technology to my patients and why I chose it for my own family.
Are you ready to learn more about how FDA-approved ortho-k can safely provide clear vision and control myopia for your child? Take our Candidacy Quiz [blocked] to see if your child is a good candidate, or contact us [blocked] to schedule a consultation. We’re here to answer all your questions and be your partner on this journey.
Frequently Asked Questions (FAQs)
1. Is ortho-k safe for children?
Yes, when prescribed and managed by a qualified eye care professional using FDA-approved lenses, ortho-k is considered a safe and effective option for myopia control in children. Numerous studies have demonstrated its safety, and the risk of complications is very low when proper hygiene and wearing schedules are followed. It’s a responsibility we take very seriously, with thorough training for every family.
2. What are the potential side effects of ortho-k?
The most common side effects are temporary and mild, such as initial lens awareness, dry eye symptoms, or slight glare and halos around lights, especially in the first few weeks. Serious complications like microbial keratitis are rare, with an incidence similar to other forms of overnight contact lens wear. Following your doctor’s instructions for cleaning, handling, and follow-up care is the best way to ensure a safe and comfortable experience.
3. How do I know if my eye doctor is experienced in fitting ortho-k lenses?
That’s a great question. You should look for a practitioner who is a Fellow of the International Academy of Orthokeratology and Myopia Control (FIAOMC). This designation indicates a high level of expertise and commitment to the field. Don’t be shy about asking how many patients they have fitted and what their success rate is. You can learn more on our How It Works [blocked] page or browse our other blog [blocked] articles.
4. How is ortho-k different from regular contact lenses?
Unlike regular soft contact lenses that are worn during the day to correct vision, ortho-k lenses are worn only at night while you sleep. They gently reshape the front surface of the eye (the cornea), which corrects your vision and allows for clear sight throughout the day without the need for glasses or contacts. This reshaping effect is temporary and reversible. Additionally, ortho-k is a primary method for myopia control in children, a benefit not provided by standard contact lenses.
5. Can I use my HSA or FSA for ortho-k treatment?
Absolutely. sleepSEE® myopia control is a specialty service not covered by insurance. Flexible payment options and HSA/FSA funds are accepted. We believe that financial considerations shouldn’t be a barrier to quality eye care, and we’re happy to work with you to make treatment affordable.
Last reviewed: February 2026
References
[1] Bullimore, M. A. (2017). The Safety of Soft Contact Lenses in Children. Optometry and Vision Science, 94(6), 638–646. https://journals.lww.com/optvissci/Fulltext/2017/06000/The_Safety_of_Soft_Contact_Lenses_in_Children.2.aspx [2] VanderVeen, D. K., Kraker, R. T., Pineles, S. L., & Hutchinson, A. K. (2019). Use of Orthokeratology for the Prevention of Myopic Progression in Children: A Report by the American Academy of Ophthalmology. Ophthalmology, 126(4), 623–636. https://www.aaojournal.org/article/S0161-6420(18)33073-2/fulltext
Free Download: The Science Behind Ortho-K
A plain-language summary of 12 key clinical studies on orthokeratology efficacy.
- Summary of 12 peer-reviewed clinical studies
- Myopia progression rates with/without treatment
- Ortho-K vs. atropine vs. multifocal lenses
- FDA approval history and safety data
Ortho-K Research Summary
Science & Evidence — Free PDF
sleepSEE Medical Advisory Board
sleepSEE Medical Advisory Board is a contributor to the sleepSEE Insights blog, sharing expert knowledge on myopia progression, orthokeratology (Ortho-K) and myopia control.